 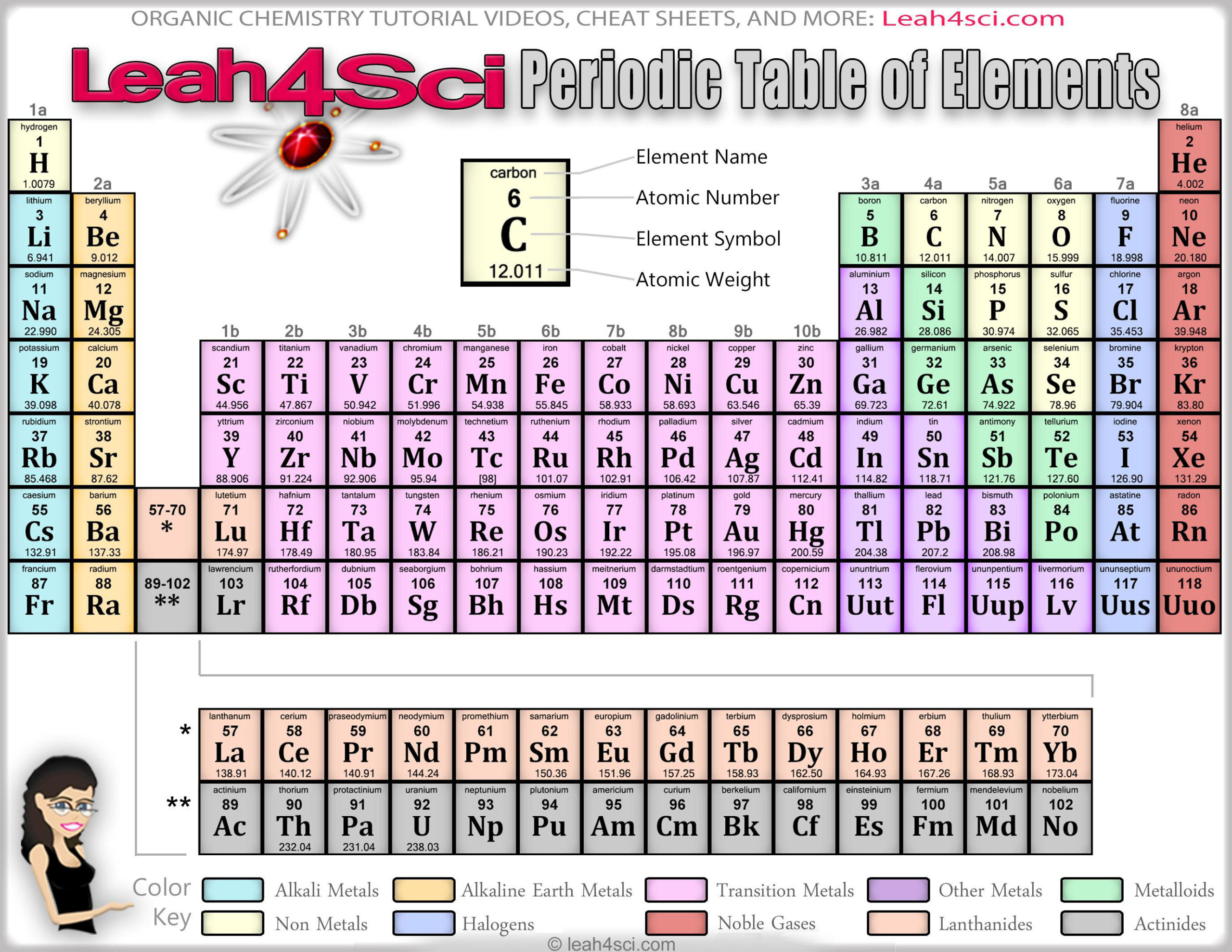
The term metalloid has also been used for elements that exhibit metallic lustre and electrical conductivity, and that are amphoteric, such as arsenic, antimony, vanadium, chromium, molybdenum, tungsten, tin, lead, and aluminium. These elements include hydrogen, beryllium, nitrogen, phosphorus, sulfur, zinc, gallium, tin, iodine, lead, bismuth, and radon. Other elements are occasionally classified as metalloids. The inclusion of antimony, polonium, and astatine as metalloids has been questioned. Sometimes tellurium is not regarded as a metalloid. Boron sometimes is excluded, by itself, or with silicon. Depending on the author, one or more from selenium, polonium, or astatine are sometimes added to the list. īoron, silicon, germanium, arsenic, antimony, and tellurium are commonly recognised as metalloids. 
Only the elements at or near the margins, lacking a sufficiently clear preponderance of either metallic or nonmetallic properties, are classified as metalloids. Most elements have a mixture of metallic and nonmetallic properties, and can be classified according to which set of properties is more pronounced. Difficulty of categorisation is a key attribute. This is a generic definition that draws on metalloid attributes consistently cited in the literature. See also: Lists of metalloids Judgment-based Ī metalloid is an element that possesses a preponderance of properties in between, or that are a mixture of, those of metals and nonmetals, and which is therefore hard to classify as either a metal or a nonmetal. In this context, only arsenic and antimony are semimetals, and commonly recognised as metalloids. In physics, it refers to a specific kind of electronic band structure of a substance. Metalloids are sometimes called semimetals, a practice that has been discouraged, as the term semimetal has a different meaning in physics than in chemistry. Its more recent meaning, as a category of elements with intermediate or hybrid properties, became widespread in 1940–1960. The term metalloid originally referred to nonmetals. The electrical properties of silicon and germanium enabled the establishment of the semiconductor industry in the 1950s and the development of solid-state electronics from the early 1960s. They and their compounds are used in alloys, biological agents, catalysts, flame retardants, glasses, optical storage and optoelectronics, pyrotechnics, semiconductors, and electronics. Metalloids are usually too brittle to have any structural uses. Most of their other physical properties and chemical properties are intermediate in nature. Chemically, they behave mostly as nonmetals. Typical metalloids have a metallic appearance, but they are brittle and only fair conductors of electricity. Some periodic tables include a dividing line between metals and nonmetals, and the metalloids may be found close to this line. On a standard periodic table, all eleven elements are in a diagonal region of the p-block extending from boron at the upper left to astatine at lower right. Five elements are less frequently so classified: carbon, aluminium, selenium, polonium and astatine. The six commonly recognised metalloids are boron, silicon, germanium, arsenic, antimony and tellurium. Despite the lack of specificity, the term remains in use in the literature of chemistry. There is no standard definition of a metalloid and no complete agreement on which elements are metalloids. A metalloid is a type of chemical element which has a preponderance of properties in between, or that are a mixture of, those of metals and nonmetals. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
